COVID-19 Vaccine Update: Legal & Practical Considerations for Employers
What does the COVID-19 vaccine mean for my employees?
Can I mandate the COVID-19 vaccine for my employees?
Get the answers to these frequently asked questions in the article below. For the full presentation, including technical information on the COVID-19 vaccines and legal and practical consideration in Canada, China, Mexico and Germany, please download the slides.
This presentation was developed by the professionals from Faegre Drinker Biddle & Reath for a webinar held on Jan. 21, 2021.
This presentation is for informational purposes only. It is not intended as legal advice for any specific situations and will not be updated after it has been distributed.
Watch the Webinar Download the Slides
Can U.S. Employers Require COVID-19 Vaccination of All Employees?
There are two things you need consider when making this decision.
- Your exposure risk level.
- Accommodations for medical contraindications, including due to pregnancy or breastfeeding, and religious objection.
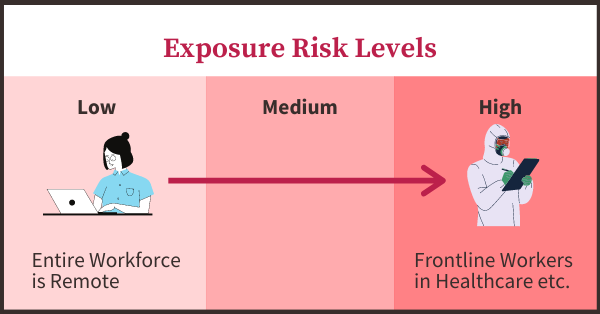
Exposure Risk Levels
Scenarios in the middle of the risk spectrum could include things like onsite workers in essential industries with high absenteeism, close-contact jobs or jobs requiring significant travel.
The less the exposure risk that cannot be addressed by other measures, the greater the risk of a mandatory vaccination requirement. For many or most employers, the analysis should be by job responsibilities.

Accommodation Obligations
CDC guidance (prevaccination checklist) as of Jan. 12, 2021, its screening questions cover:
Current sickness; History of allergic reaction; Weakened immune system caused by something such as HIV infection or cancer; Bleeding disorder or taking a blood thinner.
Not necessarily disqualifying; instructions provide guidance for HCPs
The duty to reasonably accommodate should follow a standard process:
- Additional mitigating measures such as greater distancing?
- Work from home?
- Leave of absence?
- Reassignment to an open and available position?
Employers may require health care provider verification of restrictions (treated confidentially).

Pregnancy or breastfeeding
A “healthy pregnancy” is not a disability, but a number of U.S. states and localities require accommodation of pregnancy-related restrictions in a manner similar to disability-related restrictions.
Current CDC guidance states that there is limited data available on the safety of COVID-19 vaccines administered during pregnancy and no data on the safety of the vaccines for lactating women, their breastfeeding infants, or milk production/excretion.
Faith-based Objections
Employers must accommodate short of undue hardship — defined as more than de minimis cost or burden. Any request for supporting evidence of the faith-based grounds for objection must be based on an objective reason for questioning the religious nature or sincerity of a particular belief, practice or observance.
The definition of religion is broad and may extend to belief systems not widely viewed as organized religions. For example, one federal district court has held that veganism might reflect “a sincerity equating that of traditional religious views.”

ADA Considerations
The Americans with Disabilities Act (ADA) allows qualification standards (such as vaccination) to prevent direct threats to health and safety. Because a vaccination requirement would screen out individuals with disabilities at a higher rate than others, the employer must be prepared to show that the risk cannot be sufficiently mitigated by other measures (masking, distancing, installing barriers, remote-work arrangements, etc.)
May U.S. Employers Require Proof of Vaccination, Absent an Exemption?
The Equal Employment Opportunity Commission (EEOC) says yes — merely requesting proof of vaccination is not a restricted disability-related inquiry. But prescreening inquiries such as allergic reaction histories are disability-related inquiries and must be job related and consistent with business necessity.
The employer is responsible for compliance with this requirement even if it outsources its vaccination process. Questions about family medical history could also run afoul of the Genetic Information Nondiscrimination Act (GINA). Inquiries about why someone did not get vaccinated may also be disability-related inquiries.
What Other Considerations are Important in Decision Making on Mandatory Vaccination?
- Employees who believe vaccination is unsafe may submit complaints to OSHA or state OSH agencies, despite FDA authorization for use.
- Suspected adverse side effects or later complications could lead to workers’ compensation claims and OSHA recordable injuries.
- The administration process may require significant resources.
- Confidentiality must be observed — including as to who has been vaccinated.
- Be on the lookout for any federal, state, or local legislation that may ban or require employer vaccination requirements.
- Consider a carrot rather than a stick, with wellness incentives (consider applicable incentive limits, accommodation requirements and tax considerations).

Can U.K. Employers Mandate that Their Workforce Receives the Vaccine?
In the U.K., employers have a duty to ensure the health and safety of their workforce as far as reasonably possible, but a policy of mandatory vaccination could have legal implications so employers in the U.K. should be extremely cautious.
Legal Implications of a Mandatory Vaccination Policy in the U.K.
A mandatory vaccination policy carries a risk of discrimination and/or constructive dismissal claims if an employee is treated less favourably because they refuse to be vaccinated and such refusal is related to a characteristic protected under U.K. discrimination law. Mandatory vaccination could also infringe employees’ right to privacy under article 8 of the U.K. Human Rights Act 1998. Employers should consider the data protection implications of processing information about whether employees have had the vaccine since the U.K. has strict rules on processing of health data.
Instead of mandatory vaccination, employers could introduce a vaccine policy to encourage employees to receive the vaccine. Employers should also continue to consider other protective measures.
Authors
M. Angella Castille
 Angie Castille, partner at Faegre Drinker Biddle & Reath, serves as a seasoned international general counsel partnering with companies to plan and implement their international growth, transactions and trade. For those doing business in emerging markets, she guides them through the maze of managing risk, resolving challenges and operating ethically in those jurisdictions. Angie co-chairs Faegre Drinker’s firmwide international team, a cross-disciplinary group of lawyers and consultants who advise clients on international legal and regulatory issues.
Angie Castille, partner at Faegre Drinker Biddle & Reath, serves as a seasoned international general counsel partnering with companies to plan and implement their international growth, transactions and trade. For those doing business in emerging markets, she guides them through the maze of managing risk, resolving challenges and operating ethically in those jurisdictions. Angie co-chairs Faegre Drinker’s firmwide international team, a cross-disciplinary group of lawyers and consultants who advise clients on international legal and regulatory issues.
Susan W. Kline
 A former human resources function leader and employment litigator with trial experience, Susan Kline is dedicated to helping employers take practical, effective and balanced action in HR legal matters to promote the organization’s overall business interests. As a partner at Faegre Drinker Biddle & Reath, Susan helps clients manage employment-related matters by providing counseling, training, and policy advice and review services on issues such as leave and disability management, workforce restructurings, human resources processes, employee discipline and discharge, and employment agreements.
A former human resources function leader and employment litigator with trial experience, Susan Kline is dedicated to helping employers take practical, effective and balanced action in HR legal matters to promote the organization’s overall business interests. As a partner at Faegre Drinker Biddle & Reath, Susan helps clients manage employment-related matters by providing counseling, training, and policy advice and review services on issues such as leave and disability management, workforce restructurings, human resources processes, employee discipline and discharge, and employment agreements.
Charlotte Marshall
 At Faegre Drinker Biddle & Reath, Charlotte Marshall advises businesses on a range of labor and employment issues in the U.K., including redundancies and business reorganisations; terminations and post-termination restrictions; effective workforce management; and employment contracts and related documentation. Charlotte also has experience advising employers on employment-related litigation, including defending claims of unfair dismissal, discrimination and whistle-blowing, and frequently advises businesses on the U.K. employment aspects of corporate transactions involving multinational parties.
At Faegre Drinker Biddle & Reath, Charlotte Marshall advises businesses on a range of labor and employment issues in the U.K., including redundancies and business reorganisations; terminations and post-termination restrictions; effective workforce management; and employment contracts and related documentation. Charlotte also has experience advising employers on employment-related litigation, including defending claims of unfair dismissal, discrimination and whistle-blowing, and frequently advises businesses on the U.K. employment aspects of corporate transactions involving multinational parties.
James Vergis, Ph.D.
James Vergis is a multidisciplinary Ph.D. scientist who provides scientific consulting and management support to pharmaceutical, biotechnology and medical device industry clients and serves as co-leader for the Faegre Drinker Biddle & Reath COVID-19 Task Force. He is particularly adept at helping clients streamline the development of new scientific technology and data-sharing processes. A researcher whose work has touched on various fields of study, Jamie is passionate about helping clients cultivate new scientific knowledge and drive public policy changes that enhance the drug-development process.
Pingback from aibinternational.com
Food First Blog | Is the Delta Variant Hijacking Your Return to thttps://www.aibinternational.com/en/Food-First-Blog/PostId/1434/is-the-delta-variant-hijacking-your-return-to-the-office-5-critical-tips-for-the-covid-19-delta-variant-surge


